Breadcrumb
- Home
- Research
Research
The cochlea (hearing organ) contains a heterogeneous population of resident macrophages with tissue- and niche-specific functions that support cochlear homeostasis and immune surveillance. Macrophages make up approximately 95% of the immune cells in the cochlea, making them the major immune cell type in this tissue. Two major tissue-resident macrophage populations have been described: cochlear macrophages and perivascular macrophages.

Cochlear macrophages are distributed throughout multiple cochlear compartments, including the spiral ligament, spiral limbus, organ of Corti region, and modiolus. These cells exhibit ramified morphologies with dynamic processes, suggesting an active role in environmental sensing and tissue maintenance. Under homeostatic conditions, cochlear macrophages contribute to debris clearance, regulation of local inflammation, and support of cochlear cellular integrity. In response to stressors such as noise exposure, ototoxic drugs, infection, or aging, cochlear macrophages actively participate in inflammatory and reparative responses that influence sensory cell survival and cochlear function.

Perivascular macrophages (PVMs) represent a specialized subset of cochlear macrophages that are closely associated with the cochlear vasculature, particularly within the stria vascularis. Perivascular macrophages are thought to play a critical role in immune surveillance and regulating the permeability of the blood–labyrinth barrier (BLB). Through immune surveillance and signaling interactions, they may modulate vascular inflammation, leukocyte trafficking, and fluid and ion homeostasis essential for normal auditory transduction.
Macrophages can be a double-edged sword. In some situations, they help protect and repair tissue, while in others, they can contribute to tissue damage and exacerbate organ dysfunction.
In the Sung Lab, we study different models of cochlear injury to determine whether macrophages are beneficial or harmful and to understand the mechanisms by which they influence hearing. To address this, we use powerful methods including auditory testing (ABR and DPOAE), pharmacology, single cell/nucleus RNA sequencing (scRNA-seq/snRNA-seq), immunohistochemistry, flow cytometry, Inductively Coupled Plasma Mass Spectrometry (ICP-MS), and viral transduction. Combined with genetically engineered mice, these approaches have created exciting opportunities to uncover the cellular and molecular mechanisms underlying cochlear injury and immune responses.
We are currently interested in understanding the mechanisms underlying macrophage function in hearing loss caused by cisplatin. Cisplatin is a chemotherapeutic drug that is highly effective in treating a variety of cancers. However, it is the most ototoxic drug in clinical use, causing mechanosensory hair cell death in the cochlea and primarily causing high-frequency sensorineural hearing loss that can extend to lower frequencies at higher doses. This leads to significant and permanent hearing loss in both pediatric and adult cancer patients, substantially impairing quality of life.
Recently we have identified that macrophage ablation via PLX3397 provides complete protection against cisplatin-induced hearing loss and outer hair cell death in mice, suggesting that macrophages contribute to hearing loss in cisplatin-treated mice.
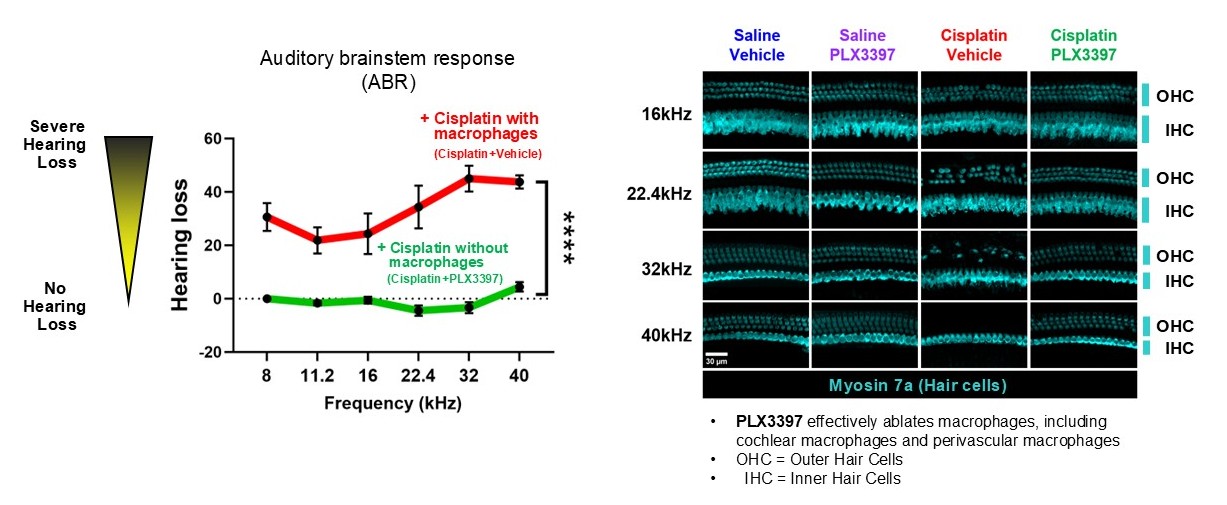
Based on these findings, we are combining the approaches described above to address the following questions:
- How do macrophages contribute to hair cell death and hearing loss?
- What macrophage subpopulations are involved in cisplatin-induced cochlear injury?
- How does cisplatin exposure alter macrophage behavior and function?
- Can modulation of macrophage activity prevent or mitigate hearing loss?
Additionally, cisplatin is administered exclusively to cancer patients in the clinical setting. The tumor microenvironment disrupts tissue homeostasis and induces dynamic alterations in the metabolism and function of stromal and immune cells, creating a biological state distinct from that of healthy mice. To complement our studies in non–tumor-bearing mice, we are therefore using a tumor-bearing mouse model we have developed to evaluate the role of macrophages in cisplatin-induced hearing loss.
Coupled with our experimental approaches, we are leveraging high-throughput single-cell and single-nucleus transcriptomics (snRNA-seq and scRNA-seq) alongside bioinformatic analyses to dissect gene expression patterns and regulatory networks underlying cochlear responses to injury. We are currently assessing differences in cochlear gene expression between control and cisplatin-treated mice and identifying key genes and pathways that may contribute to cell-specific susceptibility (or resilience) to cisplatin, using our large snRNA-seq dataset and bioinformatics tools, with a focus on immune and glial cell populations within the cochlea.
Together, by combining experimental and computational approaches, these integrative analyses will help advance our understanding of the cellular and molecular mechanisms underlying ototoxic drug-induced cochlear injury, define the roles of immune and glial cells during cochlear damage, and help guide the development of targeted protective strategies.
Funding Sources
